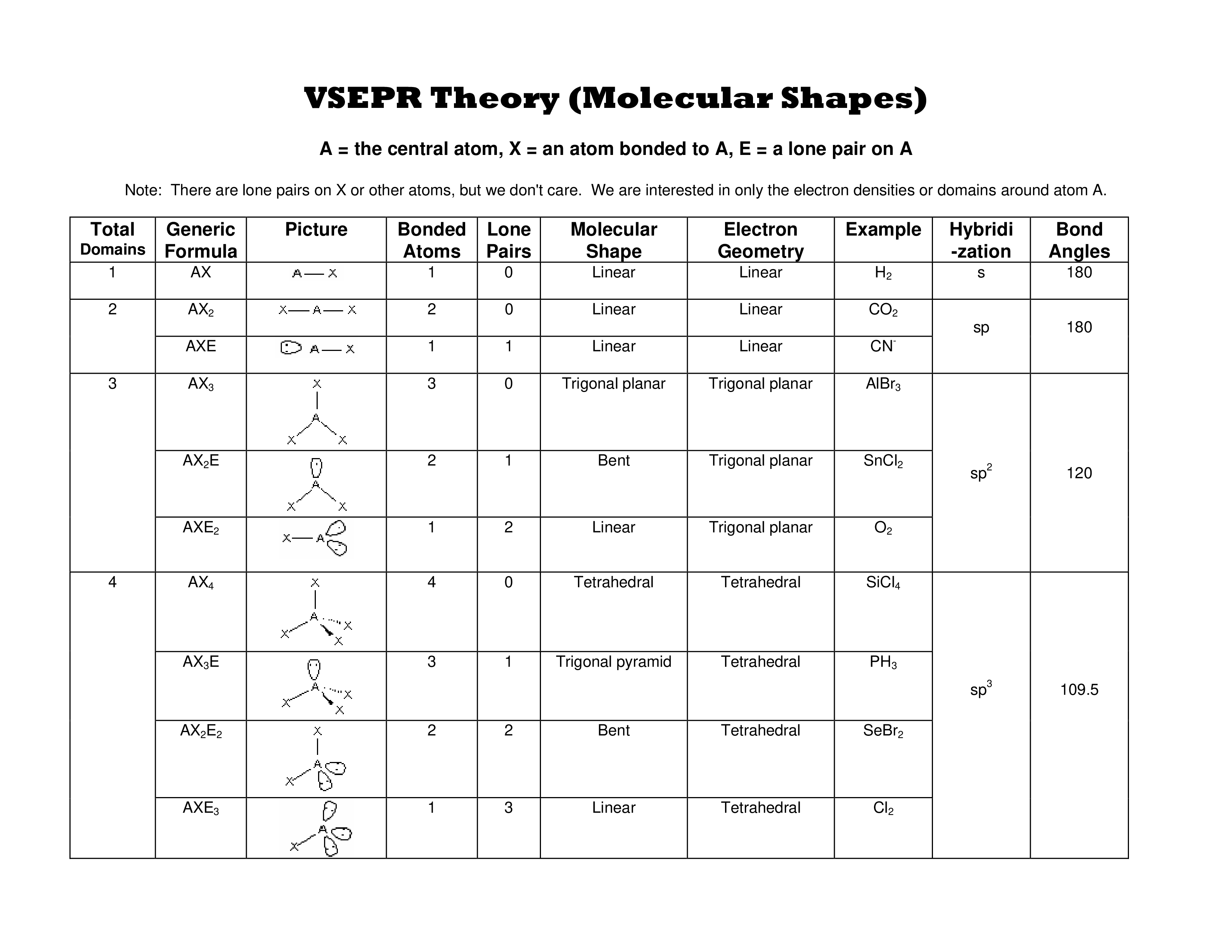
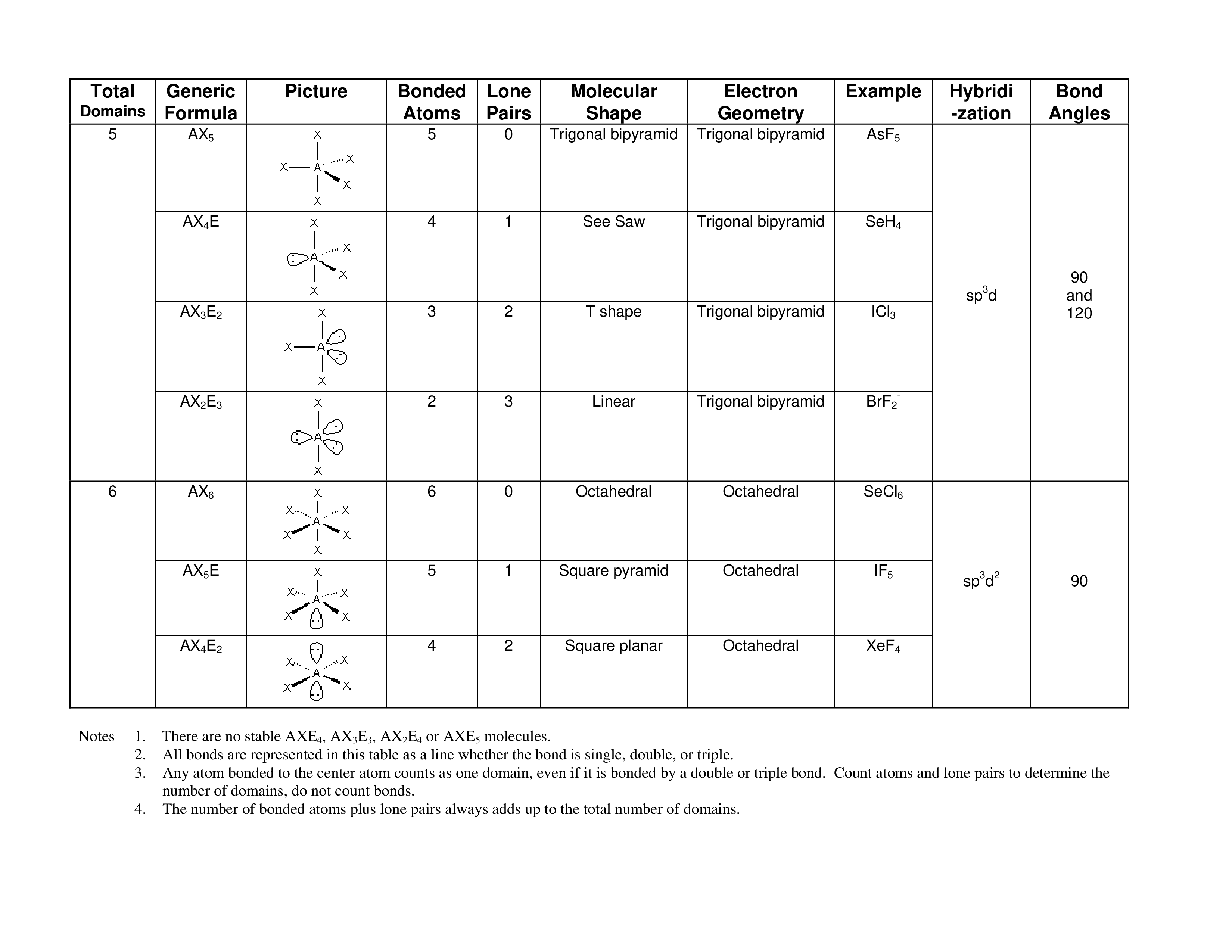
Vsepr Theory Chart
Review Rating Score
Knowing the geometry of molecules is vital to understand its reactions. VESPR is a short for Valence Shell Electron Pair Repulsion. It provides a simple method of predicting the geometries of main group compounds. The VESPR theory is applied when predicting the combinations of electron doubles around atoms in simple and symmetric molecules. In them, central atoms attach to two or more other atoms. The shapes of these key atoms and the non-bonding electron doubles influence the shape of the larger whole.
The VESPR Shape Chart available for download consists out of columns and rows. The description each of the columns and their usage is described below:
- Total domains.This column contains numbers from 1 to 6 (also called "numbers of electron domains" or "steric numbers"). A steric number is usually indicated as SN. To calculate this number, you should sum up X and E that atom A has in a molecule. Electron domains include bonded atoms, lone pairs of electrons, and unpaired electrons. You should consider multiple connections as one electron domain.
- Generic formula. It reflects the electron pairs circling a central atom. The basic formula is AxnEm, where A is the central atom, X denotes atoms circling A, and E denotes lone pairs of electrons on the A (central atom).
- A sample picture of electron-group shape represents the appearance of the molecule.
- Bonded atoms. This column contains the amount of connected atoms. To calculate this number, you should summarize all the X in the generic formula.
- Lone pairs. This column represents the amount of lone pairs, which is a place with non-bonding electrons. To calculate this number, you should summarize all the E in the generic formula.
- Molecular shape. Linear,trigonal planar tetrahedral, trigonal bipryamidal, octahedral.
- Electron geometry. This column contains the general molecular shape and other parameters, such as lengths and angles of bonds that demonstrate the location of each atom.
- An example of the compound. For example, H2, CO2, AIBR3, SnCI2, O2, PH3, and CI2
- Hybridization. This column contains hybridization. If you may determine the angles between hybrid orbitals, it will help to explain to you a molecule shape, because these angles are nearly equal to the angles between bonds. There are several types of hybridization: sp, sp2, sp3.
- Bond angles. A bond angle is a theoretical angle between two adjoining bonds. It differs depending on the shape of the molecule.
Is the template content above helpful?
Thanks for letting us know!
Reviews
Nick Porter(12/6/2021)
Quick and your not trying to scam me or ask for an email. Thanks!
Anibal Hoover(10/13/2021) - DEU
I found the right file and now I bookmarked it and will use it more
Robyn Bates(10/13/2021) - AUS
Great doc
Ophelia Roberts(10/13/2021) - DEU
Thank you!!
Winnifred Keith(10/13/2021) - GBR
Thank you for this!!
Clelia Riggs(10/13/2021) - USA
Grateful!!
Alonzo Irwin(10/13/2021) - GBR
I'm very busy, and this is very helpful
Frequently Asked Questions
What is the VSEPR theory?
What are alternative names for VSEPR Chart?
- vsepr theory chart
- vsepr geometry chart
- vsepr model chart
- vsepr table
Last modified
Our Latest Blog
- The Importance of Vehicle Inspections in Rent-to-Own Car Agreements
- Setting Up Your E-mail Marketing for Your Business: The Blueprint to Skyrocketing Engagement and Sales
- The Power of Document Templates: Enhancing Efficiency and Streamlining Workflows
- Writing a Great Resume: Tips from a Professional Resume Writer
Template Tags
Need help?
We are standing by to assist you. Please keep in mind we are not licensed attorneys and cannot address any legal related questions.
-
Chat
Online - Email
Send a message
You May Also Like
Create Professional Project Flow Charts with our Free Template
Social Event Flow Chart Template - Plan, Organize, and Execute Memorable Events
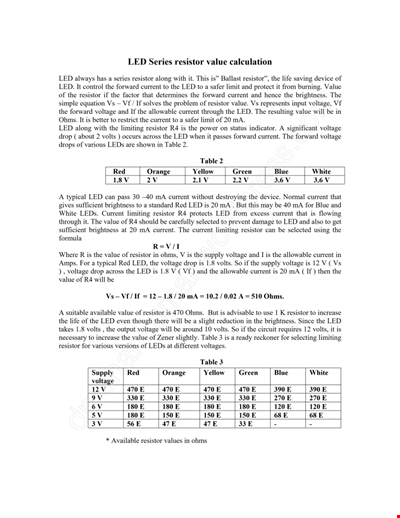
Calculation Resistor Value Chart For Led

Reward Template for Kids | Printable Behavior Chart & Chore Chart

Organizational Chart Template, Editable Org Chart, Free Download

Minecraft Birthday Banner - Buy Customizable Party Decorations
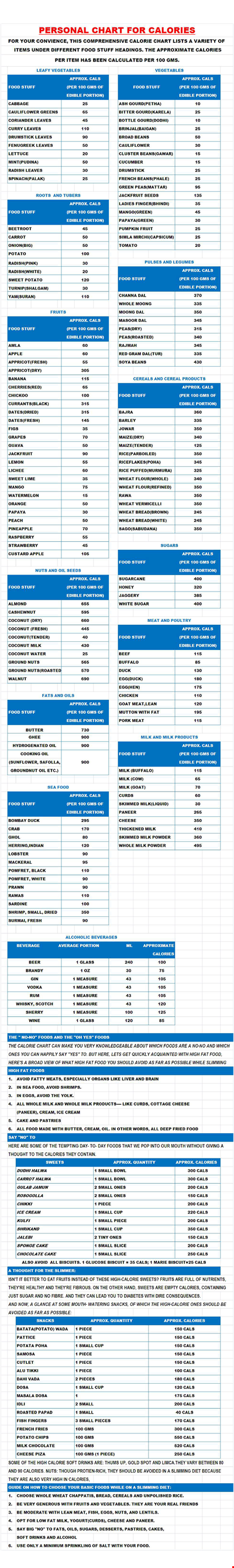
Personal Food Calorie Chart

Family Tree Chart For Kids
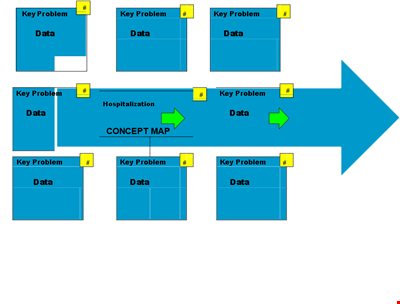
Create Organized Concepts with Our Concept Map Template - Solve Problems & Analyze Reasons
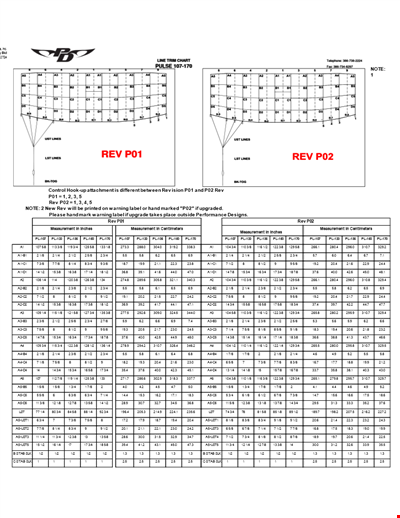
Find the Ideal Pulse Rate with Our Chord Tool
Org Chart Template Word
Understand and Reduce Process Variation with a Pareto Chart
Create an Organizational Chart Template - Easily Visualize Your Company Hierarchy

Free Vintage Family Tree Template
Large Hospital Organizational Chart Template - Download Now

Printable Shoe Size Chart for Kids: Find the Perfect Fit for Your Little Ones